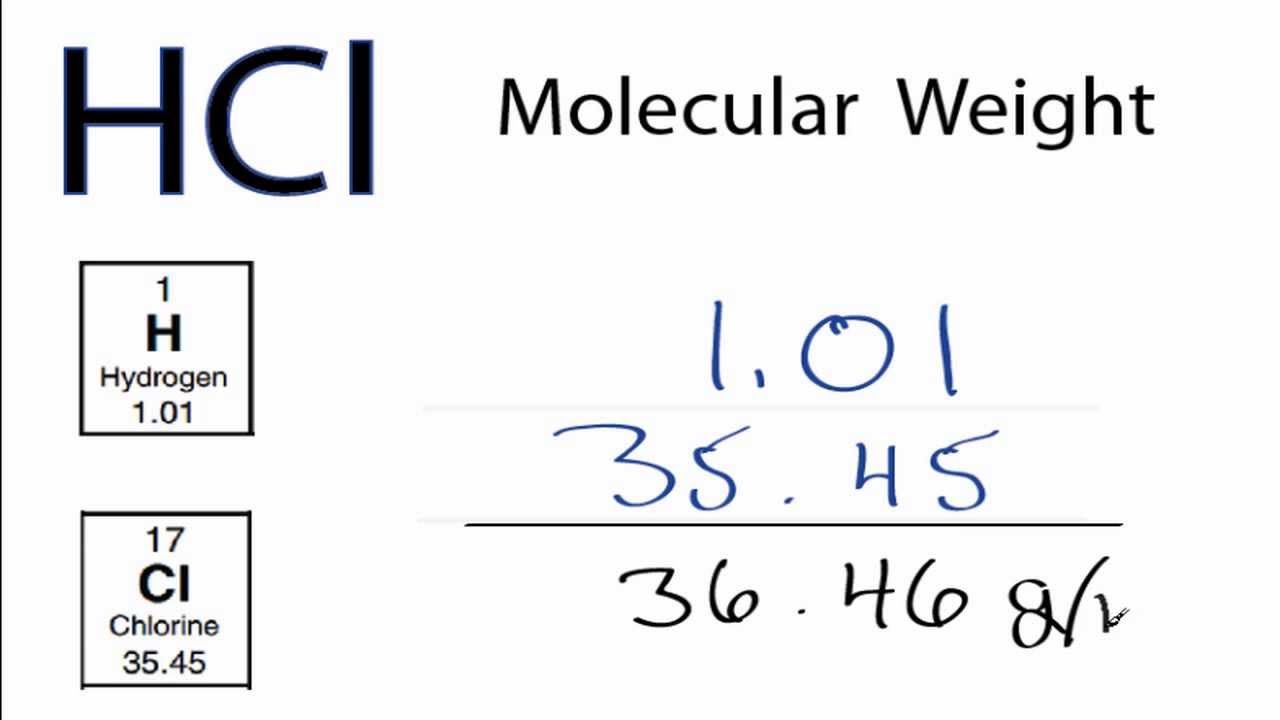
So the molecular weight of Ammonium nitrate is 80.04368. Step 4: Calculate the molecular weight of Ammonium nitrate by adding up the total weight of all atoms. Total weight of Oxygen atoms in Ammonium nitrate: 15.9994 x 3 = 47.9982 Number of Oxygen atoms in Ammonium nitrate: 3 
Total weight of Hydrogen atoms in Ammonium nitrate: 1.008 x 4 = 4.032 Number of Hydrogen atoms in Ammonium nitrate: 4 Total weight of Nitrogen atoms in Ammonium nitrate: 14.00674 x 2 = 28.01348 Number of Nitrogen atoms in Ammonium nitrate: 2 Step 3: Calculate the total weight of each atom in an Ammonium nitrate molecule by multiplying its atomic weight by its number. Step 2: Find out the atomic weights of each atom (from the periodic table).Ītomic weight of Nitrogen (N): 14.00674 (Ref: Jlab-ele007)Ītomic weight of Hydrogen (H): 1.008 (Ref: Lanl-1)Ītomic weight of Oxygen (O): 15.9994 (Ref: Jlab-ele008) From the chemical formula of Ammonium nitrate, you can find that one molecule of Ammonium nitrate has two Nitrogen (N) atoms, four Hydrogen (H) atoms, and three Oxygen (O) atoms. The chemical formula of Ammonium nitrate is N 2H 4O 3. Saliva and urine were analyzed for volatile N-nitrosamines, nitrate & nitrite.Blood was analyzed for nitrate. Every hydrogen atom has one proton in its nucleus. 
Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an elements atomic number is indicated above the elemental symbol. Samples of blood, saliva & urine were collected just before a 24 hr period. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. You will know different atoms and their number in an Ammonium nitrate molecule from the chemical formula. 12 Healthy volunteers ingested orally 7-10.5 g ammonium nitrate in a single dose & 12 others received 9.5 g of sodium nitrate iv in approx 60 min. Step 1: Find the chemical formula and determine constituent atoms and their number in an Ammonium nitrate molecule. The molecular weight of Ammonium nitrate is 80.04368 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of Nitrogen, Hydrogen, and Oxygen.ĬALCULATION PROCEDURE: Ammonium nitrate Molecular Weight Calculation The photons emitted by caesium are more energetic than these of lithium.The molecular weight of Ammonium nitrate is 80.04368.Īmmonium nitrate is an inorganic compound of three elements: Nitrogen, Hydrogen, and Oxygen. It corresponds to a decrease of the wavelength of the light emitted during the electrons de-excitation step in the atoms brought at high temperature. In a general way, the emitted color progressively turns from the red to the violet in the visible spectrum of light when going down in the column of the alkaline metals in the periodic table of Mendeleev.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
